L型钙离子通道(CaV1)的调控方法,尤其是通道抑制剂,一直是基础研究和药物开发的热点领域💺。作为重要的电压门控离子通道,CaV1在多种生理功能中发挥关键作用🤜🏽,包括肌肉收缩、学习记忆和激素分泌等🧑🏻🦳。因此,通道抑制方法的研究不仅对深入理解其分子生理学具有重要意义,还可以为治疗高血压、心律失常和神经退行性疾病等提供新的靶点和手段。与其他传统小分子抑制剂相比👩🍳,基因编码的碳末端介导抑制(CMI)多肽在可控性🐕🦺、特异性🚴🏼♀️📇、适用性和有效性等方面具有潜在优势🥴,然而在其应用推广过程中一直受限于若干瓶颈问题,例如👩🏿🎤:时-空可控精度亟待提高、原代细胞内源性钙通道复杂性(存在多种亚型及变体)导致的动态空间困境等。
11月18日🚣🏼♂️,Cell Press(细胞出版社)期刊《Cell Reports Methods》在线发表题为“Opto-chemogenetic inhibition of L-type CaV1 channels in neurons through a membrane-assisted molecular linkage”的研究论文🧣。该工作由凯发娱乐刘晓冬教授课题组联合樊瑜波教授团队与合作者共同完成。该工作通过整合近红外(NIR)敏感纳米粒子👰♂️、雷帕霉素(rapamycin)诱导二聚化、以及Ras/膜辅助的类分子内链接,实现对神经元内源性CaV1通道的高效特异性抑制。基于重组通道开发的抑制肽等分子元件递送至细胞𓀗,在近红外光诱导下以高时空分辨率抑制神经元钙通道活动—突起生长耦联,为研究神经元钙通道-钙信号的相关机制提供了新型分子工具👨🏼🔧,展现出广泛的应用潜力🙎🏻♂️。

本工作首先报道了一种膜辅助的类分子连接效应🍣,结合此前钙通道碳末端的机制研究(Yang et al. Communications Biology 2022),提出了面向内源性钙通道的CMI抑制肽设计方案。研究人员通过近红外光控制纳米粒子释放雷帕霉素诱导FRB/FKBP的结合,促进在细胞质中的抑制肽转移到细胞膜上,迅速、显著并特异性地抑制钙通道的活性👇。利用自主开发的钙探针GCaMP-X(Geng et al. eLife 2022),研究人员证实,CMI抑制肽能够响应近红外光下调神经元钙振荡-突起发育耦联🩸,为CaV1通道的生理功能提供了强有力的直接证据👨👩👧👧。本文提出的光控CMI抑制肽为“光基因生理学”又增添一类新工具,可实现秒-分钟及单细胞-亚细胞级别的高精度调控;同时,借助近红外光的强组织穿透性💁🍖,有望在活体动物水平上实现对CaV1通道及其病生理过程的精准干预。
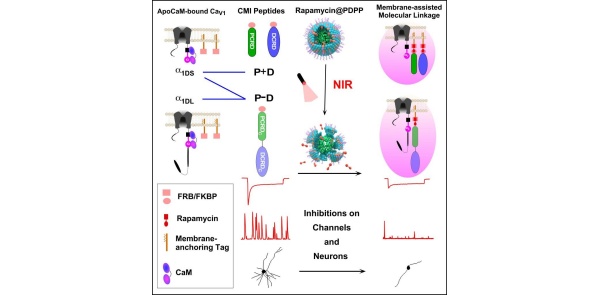
靶向神经元L型钙通道的光-化学可控抑制多肽
北京凯发K8娱乐平台注册官方网站为本工作的第一完成单位👨👧。论文的共同第一作者为北京凯发K8娱乐平台注册官方网站博士生耿金丽🎼、北京凯发K8娱乐平台注册官方网站杨亚雄副教授、河北工业大学博士后李博颖、北京凯发K8娱乐平台注册官方网站博士生于振、邱爽。此外🫡,刘楠、刘艺、王波🤽♀️、张雯及高世欣等课题组成员也为本研究做出了贡献🧑🦼➡️。本工作的通讯作者是刘晓冬教授(北航)、邢成芬教授(河北工业大学)及樊瑜波教授(北航)。本研究得到了国家自然科学基金🧖🏻♂️、中国博士后基金及北京市生物医学工程高精尖创新中心的支持。
全文链接:https://doi.org/10.1016/j.crmeth.2024.100898
Cell Reports Methods | Liu Lab Publishes New Method for Opto-chemogenetic Inhibition of Calcium Channels in Neurons
Inhibition of L-type calcium channels (CaV1) remains a critical focus in basic research and therapeutics development. As a major subfamily of voltage-gated calcium channels, CaV1 channels serve as a signaling hub for physiological functions, including muscle contraction, learning and memory, and vesicle secretion. Thus, novel methods for channel inhibition are crucial for advancing molecular physiology and developing therapeutics for cardiovascular and neurodegenerative diseases. Genetically encoded peptides that perform C-terminus-mediated inhibition (CMI) offer enhanced controllability, specificity, and efficiency compared to small-molecule and other conventional inhibitors. However, current CMI designs face challenges such as limited spatiotemporal control and the complexity of calcium channel subtypes and variants.
On November 18, 2024, the research article "Opto-chemogenetic inhibition of L-type CaV1 channels in neurons through a membrane-assisted molecular linkage" was published in Cell Reports Methods, a Cell Press journal. Led by Prof. Xiaodong Liu's lab, in collaboration with Prof. Yubo Fan from Beihang University, this study presents a novel approach integrating near-infrared (NIR)-responsive nanoparticles, rapamycin-induced dimerization, and Ras/membrane-assisted molecular linkage to achieve efficient, specific inhibition of neuronal CaV1 channels. The study designed and validated inhibitory peptides for both recombinant and endogenous CaV1 channels. Using NIR, the researchers achieved precise inhibition of Ca²⁺ flux-neuritogenesis coupling with high spatiotemporal resolution. This innovative approach offers a powerful tool for investigating neuronal calcium signaling and related mechanisms.

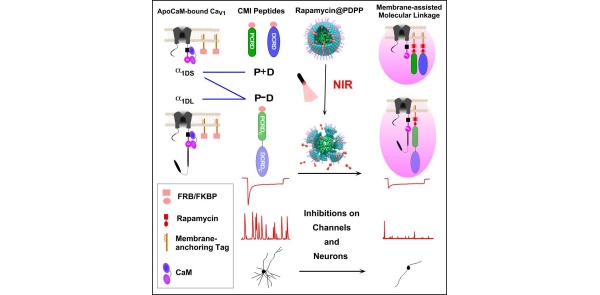
Building on previous studies (Yang et al., Communications Biology 2022), the team introduced a membrane-assisted molecular linkage for CMI peptides. NIR-triggered rapamycin release facilitated FRB/FKBP binding, driving inhibitory peptides from the cytoplasm to the membrane, resulting in rapid and specific inhibition of CaV1 activity. Utilizing GCaMP-X calcium sensors (Geng et al., eLife 2022), the researchers demonstrated the downregulation of Ca²⁺ oscillation-neuritogenesis coupling, confirming CaV1’s physiological role in neurons.
This method extends the toolkit of “optogenetic physiology,” enabling precise regulation at single-cell and subcellular levels with second-to-minute temporal resolution. With NIR’s deep tissue penetration, the approach holds promise for studying and targeting CaV1-related pathologies in vivo.
The co-first authors of this paper are Jinli Geng (Beihang University), associate professor Yaxiong Yang (Beihang University), lecturer Boying Li (Hebei University of Technology), Zhen Yu and Shuang Qiu (Beihang University). Former and present Liu-lab members including Nan Liu, Yi Liu, Bo Wang, Wen Zhang and Shixin Gao also contributed to this work. Corresponding authors of this work are Prof. Xiaodong Liu (Beihang University), Prof. Chengfen Xing (Hebei University of Technology) and Prof. Yubo Fan (Beihang University). This work is supported by the grants from the National Science Foundation of China, the China Postdoctoral Science Foundation and Advanced Innovation Center for Biomedical Engineering.
Full text links: https://doi.org/10.1016/j.crmeth.2024.100898